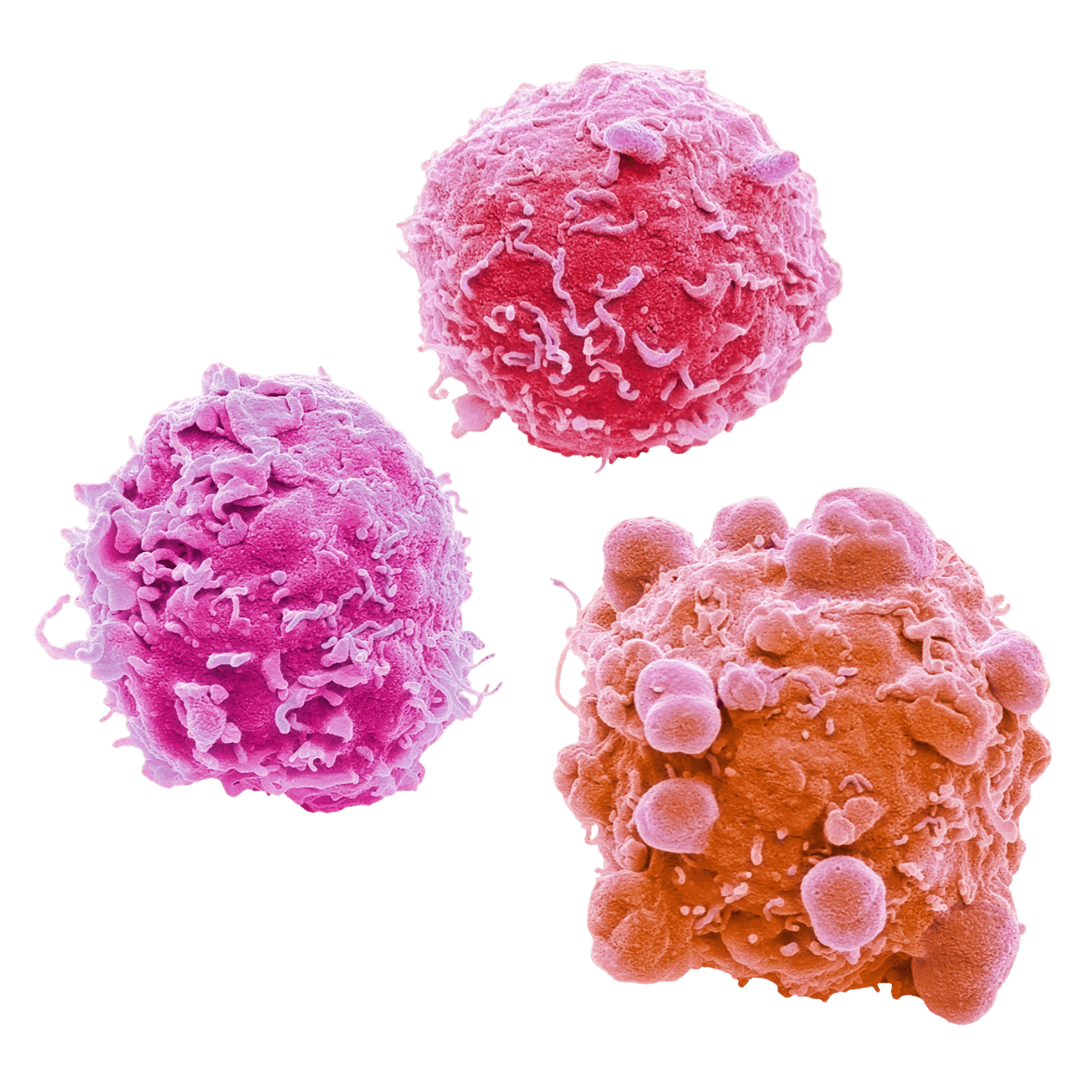
Janet DeLeon serves as President and CEO of Etienna Bio, Inc. Mrs. DeLeon has spent over 35 years working in the pharmaceutical industry. She progressed her career by assuming lead positions in Regulatory Affairs and Product Development in large and small pharmaceutical companies and consulting firms while gaining product approvals from the US FDA. For 12 years, she has been the owner and principal consultant of a regulatory and product development consulting firm developing new products from scratch. Janet has interacted with the US FDA for over 30 years by submitting, defending and gaining approval for hundreds of applications to the FDA. She was also president of PTEA, an FDA and Industry discussion group, for 12 years. Janet earned her B.S. in Chemistry from Quincy University and is certified in pharmaceutical Regulatory Affairs (RAC) by the Regulatory Affairs Professional Society.